Researchers have developed the first mathematically supported cellular map of lung tissue in idiopathic pulmonary fibrosis (IPF) and uncovered key immune cell interactions that could explain why lungs fail to repair in this deadly disease with no known cure.
The study by researchers at the University of Oxford’s MRC Translational Immune Discovery Unit, in collaboration with Newcastle University and supported by the NIHR Oxford Biomedical Research Centre (BRC), reveal a critical interaction between immune and epithelial cells that could be driving the disease — and open the door to new treatment approaches. The findings have been published in Nature Communications.
IPF is a chronic and progressive condition in which scarring builds up in the lungs, making it increasingly difficult to breathe. Although current medications can slow its progression, they do not stop or reverse the damage.
For years, scientists have known that abnormal healing processes contribute to the disease. However, the exact cellular players and how they interact in the lungs remained largely unclear.
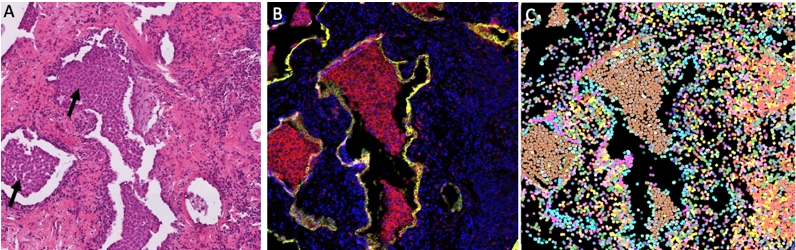
Using advanced imaging technology and computational analysis, the research team created a high-resolution ‘cellular atlas’ of lung tissue donated by IPF patients who were undergoing lung transplantation. The atlas captures both the identity and location of immune and structural cells in diseased lung tissue across different stages of the disease.
Professor Ling-Pei Ho, who led the study, said: “We’ve taken lung tissue samples and looked at them in an entirely new way — not just identifying the cell types present, but mapping where they are in relation to each other. This allows us to understand how certain immune cells may be influencing abnormal lung repair.”

Professor Ho, of the MRC Weatherall Institute for Molecular Medicine and Nuffield Department of Medicine, is the Oxford BRC’s Theme Lead for Respirator Medicine.
A major finding from the study was the discovery of a highly specific spatial link between two cell types: a damaged epithelial cell known as an aberrant basal intermediate (ABI), and a distinct immune cell called the CD206hi macrophage. These macrophages were the only immune cells found to spatially associate with ABIs at all stages of the disease.
Further analysis showed that the two cell types communicate through specific molecular signals — suggesting they may work together in a way that reinforces scarring and prevents proper lung regeneration.
“By combining spatial analysis, single-cell RNA sequencing, and protein profiling, we were able to pinpoint the immune cells that may be actively driving the faulty repair process in IPF lungs,” said Praveen Weeratunga, first author of the study. “This could help guide the development of treatments aimed at restoring healthy lung repair.”
Professor Ho added: “This study highlights the power of interdisciplinary research, bringing together experts in IPF and lung diseases, cellular imaging, immunology, pathology, computational biology and mathematics to tackle one of the most complex and challenging diseases in respiratory medicine.”
